Conventional spontaneous Raman spectroscopy of interfacial molecules typically requires plasmonic or electronic enhancement, limiting accessible systems. A nonlinear coherent Raman method now enables direct, high sensitivity detection without such requirements. A time-frequency engineered optical design suppressed strong substrate background signals by four orders of magnitude, allowing Raman detection of ångström scale ultrathin interfacial molecular layers. This method opens new opportunities for real-time operando observations in electrochemistry, catalysis, adhesion, and molecular devices.
Molecular vibrations provide direct fingerprints of chemical bonding, molecular structure, and intermolecular interactions. Raman spectroscopy is therefore one of the most powerful analytical tools in chemistry, physics, and materials science. Because it can probe vibrational modes that are difficult to access by infrared spectroscopy and can operate under ambient conditions, Raman spectroscopy has long been expected to play an important role in the study of molecular systems at surfaces and interfaces. However, direct Raman measurements of ultrathin interfacial molecular layers have remained a longstanding challenge. Ångström scale molecular films contain only a very small number of molecules, resulting in extremely weak signals. Conventional approaches therefore often rely on plasmonic field enhancement or electronic resonance. Yet these strategies are not universally applicable: plasmonic enhancement typically requires nanostructured surfaces that can modify the original interface, while resonance methods are limited to specific materials or molecules.
Researchers at the Institute for Molecular Science (NINS, Japan) and SOKENDAI have now overcome these limitations by a time-frequency engineered coherent Raman spectroscopy that enables direct and highly sensitive detection of ångström-scale molecular films without plasmonic field enhancement and electronic resonance. This approach is based on coherent Raman scattering driven by third-order nonlinear optical interactions. In coherent Raman processes, molecular vibrations are actively driven by light fields, generating directional and intense vibrational signals that can be much stronger than spontaneous Raman scattering. However, practical application of coherent Raman spectroscopy to surfaces and interfaces has been severely hindered by the presence of extremely strong nonresonant background signals generated from bulk substrates, which usually overwhelm the weak response from interfacial molecules.
To overcome this challenge, our approach combines femtosecond pump and Stokes pulses with a time delayed, asymmetrically shaped picosecond probe pulse. By precisely controlling the temporal overlap of the three excitation pulses, the instantaneous nonresonant background generated from the substrate was suppressed by approximately four orders of magnitude. Importantly, rather than eliminating the background signal completely, a controlled residual component was intentionally used as an internal local oscillator. Through optical interference with the molecular coherent Raman signal, this residual background provided more than one order of magnitude signal amplification. This strategy converts background signals from an obstacle into a useful resource for highly sensitive interfacial Raman spectroscopy.
Using this method, direct Raman spectra were successfully obtained from ångström scale molecular layers formed even on atomically flat metal surfaces with minimal surface area and without plasmonic nanostructures and electronic resonance. Associate Professor Toshiki Sugimoto, the project's principal investigator, notes that "Our approach is expected to enable versatile Raman studies of functional interfaces that have previously been difficult to access without artificial enhancement structures. We anticipate broad applications in real-time and operando analysis of electrochemical reactions, detection of reactive intermediates on catalyst surfaces, molecular and chemical characterization of adhesion interfaces, and studies of molecular and organic electronic devices."
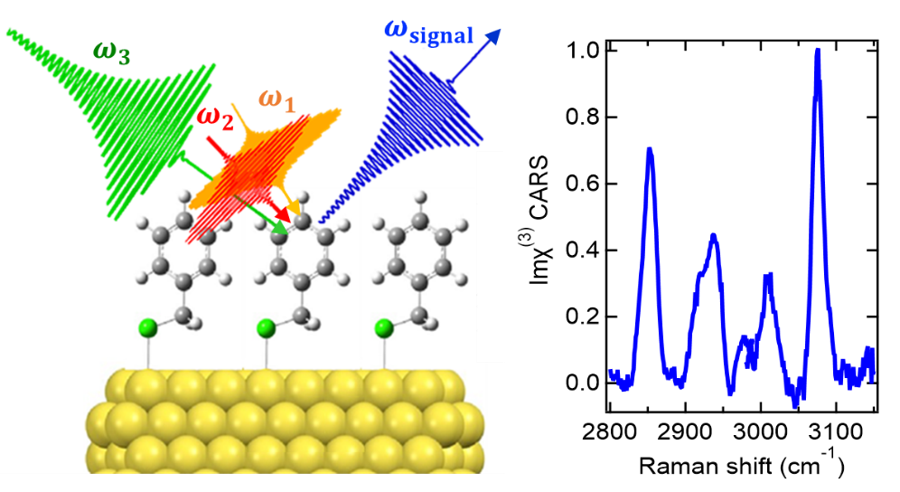
Figure 1. Experimental demonstration of plasmonic- and electronic-enhancement-free coherent Raman spectroscopy of ångström-scale ultrathin molecular systems.
(Credit: Institute for Molecular Science / Toshiki Sugimoto, Usage Restrictions: Credit must be given to the creator., License: CC BY)
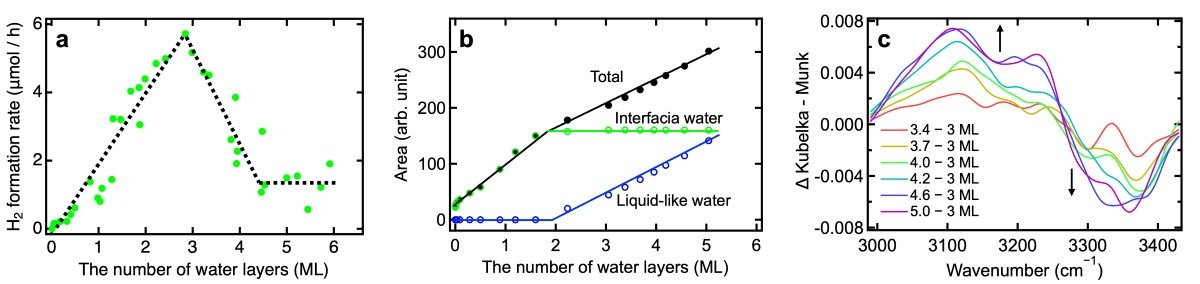
Figure 2: Impact of water adsorption on the H2 formation rate and interfacial H-bonds for the brookite TiO2 photocatalyst (TIO19PB).
(a) Change in the rate of hydrogen formation through photocatalytic water splitting when the number of water layers (amount of adsorbed water molecules) is systematically changed under various water vapor pressures. (b) The dependency of the O−H stretching band area on the number of water layers (black: total spectrum area, green: spectrum area of interfacial water component, blue: spectrum area of liquid-like water component). The spectrum of the interfacial water component, which shows a spectral shape clearly different from that of bulk liquid water, is nearly saturated in area at two molecular layers, while the liquid-like water component, which shows a spectral shape almost the same as that of bulk liquid water, increases when water molecules are adsorbed in layers thicker than three. (c) Change in the O-H vibration spectrum of the interfacial water component induced by the adsorption of more than three molecular layers of water.
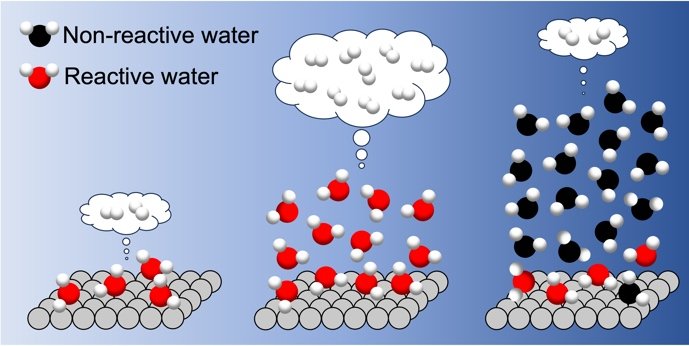
Figure 3: Summary of positive and negative impacts of interfacial hydrogen bonds on photocatalytic hydrogen evolution.
Schematic of changes in the reaction activity associated with variation of water layer thickness on the photocatalyst surface. The H2 formation rate increases with increasing water layers up to three layers. When more than three water layers cover the surface, the liquid-like water overlayers strengthen/harden the interfacial H-bond networks. The hardened interfacial H-bond networks hinder the interfacial proton-coupled hole transfer, resulting in a dramatic decrease in the H2 formation rate.
Paper Information:
Authors: Toshiki Sugimoto, Tomoaki Ichii, Tsuneto Kanai, Ryu Yoshizawa, Shota Takahashi, Atsunori Sakurai, Keisuke Seto, and Chengxiang Jin
Journal Name: Nano Letters
Journal Title: "Plasmonic- and Electronic-Enhancement-Free Coherent Raman Detection of Ångström-Scale Molecular Layers at Metal Interfaces"
DOI: 10.1021/acs.nanolett.6c00802
Financial Support:
・JSPS KAKENHI
Grant-in-Aid for Scientific Research (A), 22H00296
Grant-in-Aid for Challenging Research (Exploratory), 21K18896, 24K21759
Grant-in-Aid for Scientific Research (B), 23K26548
・ATLA Japan Innovative Science and Technology Initiative for Security, JPJ004596
・JST-FOREST, JPMJFR221U
・JST-CREST, JPMJCR22L2
・JST-K Program, JPMJKP24W1
・Amada Foundation General Research and Development Grant, AF-2021212-B2
・Joint Research by the National Institutes of Natural Sciences (NINS), 01112104
・Special Project by Institute for Molecular Science, 22IMS1101
Contact Person:
Name: Toshiki Sugimoto
TEL: +81-564-55-7280
E-mail: toshiki-sugimoto_at_ims.ac.jp (Please replace the "_at_" with @)



