Research Theme
Development of Heterogeneous Catalysis toward Ideal Chemical Processes
Keywords
Transition Metal Catalysis, Green Chemistry, Organic Synthesis
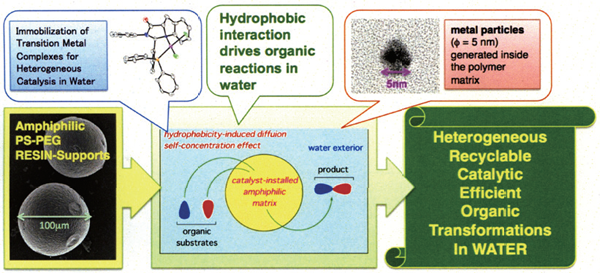
Our research interests lie in the development of transition metal-catalyzed reaction systems toward ideal (highly efficient, selective, green, safe, simple, etc.) organic transformation processes. In one active area of investigation, we are developing the heterogeneous aquacatalytic systems. Various types of catalytic organic molecular transformations, e.g. carbon-carbon bond forming cross-coupling, carbon-heteroatom bond forming reaction, aerobic alcohol oxidation, etc., were achieved in water under heterogeneous conditions by using amphiphilic polymer-supported transition metal complexes and nanoparticles, where self-concentrating behavior of hydrophobic organic substrates inside the amphiphilic polymer matrix played a key role to realize high reaction performance in water.
Selected Publications
-
Takao Osako, Kaoru Torii, Shuichi Hirata, Yasuhiro Uozumi “Chemoselective Continuous-Flow Hydrogenation of Aldehydes Catalyzed by Platinum Nanoparticles Dispersed in an Amphiphilic Resin” ACS Catal. 7, 7371-7377 (2017).
-
Go Hamasaka, Tsubasa Muto, Yoshimichi Andoh, Kazushi Fujimoto, Kenichi Kato, Masaki Takata, Susumu Okazaki, Yasuhiro Uozumi “Detailed Structural Analysis of a Self-Assembled Vesicular Amphiphilic NCN-Pincer Palladium Complex by Wide-Angle X-Ray Scattering and Molecular Dynamics Calculations” Chem. Eur. J. 23, 1291-1298 (2017).
-
Heeyoel Baek, Maki Minakawa, Yoichi M. A. Yamada, Jin Wook Han, Yasuhiro Uozumi “In-Water and Neat Batch and Continuous-Flow Direct Esteri cation and Transesteri cation by a Porous Polymeric Acid Catalyst” Sci. Rep. 6, 25925 (2016).
-
Yoichi M. A. Yamada, Yoshinari Yuyama, Takuma Sato, Shigenori Fujikawa, Yasuhiro Uozumi “A Palladium-Nanoparticle and Silicon- Nanowire-Array Hybrid: A Platform for Catalytic Heterogeneous Reactions” Angew. Chem. Int. Ed. 53, 127-131 (2014).
-
Yoichi M. A. Yamada, Shaheen M. Sarkar, Yasuhiro Uozumi “Amphiphilic Self-Assembled Polymeric Copper Catalyst to Parts per Million Levels: Click Chemistry,” J. Am. Chem. Soc. 134, 9285–9290 (2012).
-
Yoichi M. A. Yamada, Shaheen M. Sarkar, Yasuhiro Uozumi, “Self-Assembled Poly(imidazole-palladium): Highly Active, Reusable Catalyst at Parts per Million to Parts per Billion Levels,” J. Am. Chem. Soc. 134, 3190-3198 (2012).
-
Shaheen M. Sarkar, Yoichi M. A. Yamada, Yasuhiro Uozumi, “A highly Active and Reusable Self-Assembled Poly (Imidazole/Palladium) Catalyst: Allylic Arylation/Alkenylation,” Angew. Chem. Int. Ed. 50, 9437-9441 (2011).
-
Go Hamasaka, Tsubasa Muto, Yasuhiro Uozumi, “Moleular-Architecture-Based Administration of Catalysis in Water: Self-Assembly of an Amphiphilic Palladium Pincer Complex,” Angew. Chem. Int. Ed. 50, 4876-4878 (2011).
-
Yasuhiro Uozumi, Yutaka Matsuura, Takayasu Arakawa, Yoichi M. A. Yamada Asymmetric Suzuki-Miyaura Coupling in Water with a Chiral Palladium Catalyst Supported on an Amphiphilic Resin Angew. Chem. Int. Ed. 48, 2708-2710 (2009).
-
Yoichi M. A. Yamada, Takayasu Arakawa, Heiko Hocke, Yasuhiro Uozumi, “A Nanoplatinum Catalyst for Aerobic Oxidation of Alcohols in Water,” Angew. Chem. Int. Ed. 46, 704-706 (2007).
-
Yasuhiro Uozumi, Yoichi M. A. Yamada, Tomohiko Beppu, Naoshi Fukuyama, Masaharu Ueno and Takehiko Kitamori, “Instantaneous Carbon-Carbon Bond Formation Using a Microchannel Reactor with a Catalytic Membrane,” J. Am. Chem. Soc. 128, 15994-15995 (2006).
-
Shintaro Okumura, Teruki Takahashi, Kaoru Torii, Yasuhiro Uozumi, “Photocatalytic Cross-Pinacol Coupling Promoted by Carbon Dioxide” Chem. Eur. J. e202300840 (2023)
-
Shintaro Okumura, Yasuhiro Uozumi, “Photocatalytic Carbinol Cation/Anion Umpolung: Direct Addition of Aromatic Aldehydes and Ketones to Carbon Dioxide” Org. Lett. 23, 7194-7198 (2021)
-
Yoichi, M. A. Yamada, Heeyoel Baek, Takuma Sato, Aiko Nakao, Yasuhiro Uozumi, Y, “Metallically gradated silicon nanowire and palladium nanoparticle composites as robust hydrogenation catalysts” Commun. Chem. 3, 81-89 (2020)
-
Go Hamasaka, David Roy, Aya Tazawa, Yasuhiro Uozumi, “Arylation of Terminal Alkynes by Aryl Iodides Catalyzed by a Parts-per-Million Loading of Palladium Acetate” ACS Catalysis, 9, 11640-11646(2019)